Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart. Introduction to Chemical Engineering Thermodynamics. Obtain the specific volume (v) under such conditions BUY. The volumetric temperature expansion coefficient, β for water is 0. The density of water at 4☌ and 1 atm is 1000 kg/m3.In this case, a density of '1. So, density is defined as mass per unit of volume. NIST standard pressure is (101.325 kPa or 14.696 psi or 1 atm) The idea here is that you need to use the density of water at 4'C' and the volume of the sample to find its mass, then use waters molar mass to find how many moles of water you get in this sample.NIST Standard temperature is (20 C or 293.15 K or 68 F).β is the volumetric temperature expansion coefficient, β for water is 0.0002 / C°.ρ is the density of water at a specific temperature.Hence, water is denser at 4 ° C because the volume is minimum here. And we know that when volume decreases, density increases. The reason behind this is that the cage-like structure starts to break and come closer. The basic physics concept I hope almost every one might be aware of i.e ' water tend to expand instead of freezing just below and above 4 degree celsius '. The formula for the density of water based on temperature is: The temperature of the water increases and gets to 4 ° C, here, the density is maximum. However, this can be automatically converted to compatible units via the pull-down menu. The volume of a drop of water is 0.05 mL and the density of water is 1.0 g/mL. ( β) Volumetric Expansion Coefficient (default: 0.0002 / C° for water)ĭensity of Water (ρ): The calculator returns the density in kilograms per cubic meters. Click here:pointup2:to get an answer to your question :writinghand:42 the density of water at 4c is 1 gml the volume occupiedby one molecule.

INSTRUCTIONS: Choose units and enter the following: Find more Chemistry widgets in WolframAlpha.

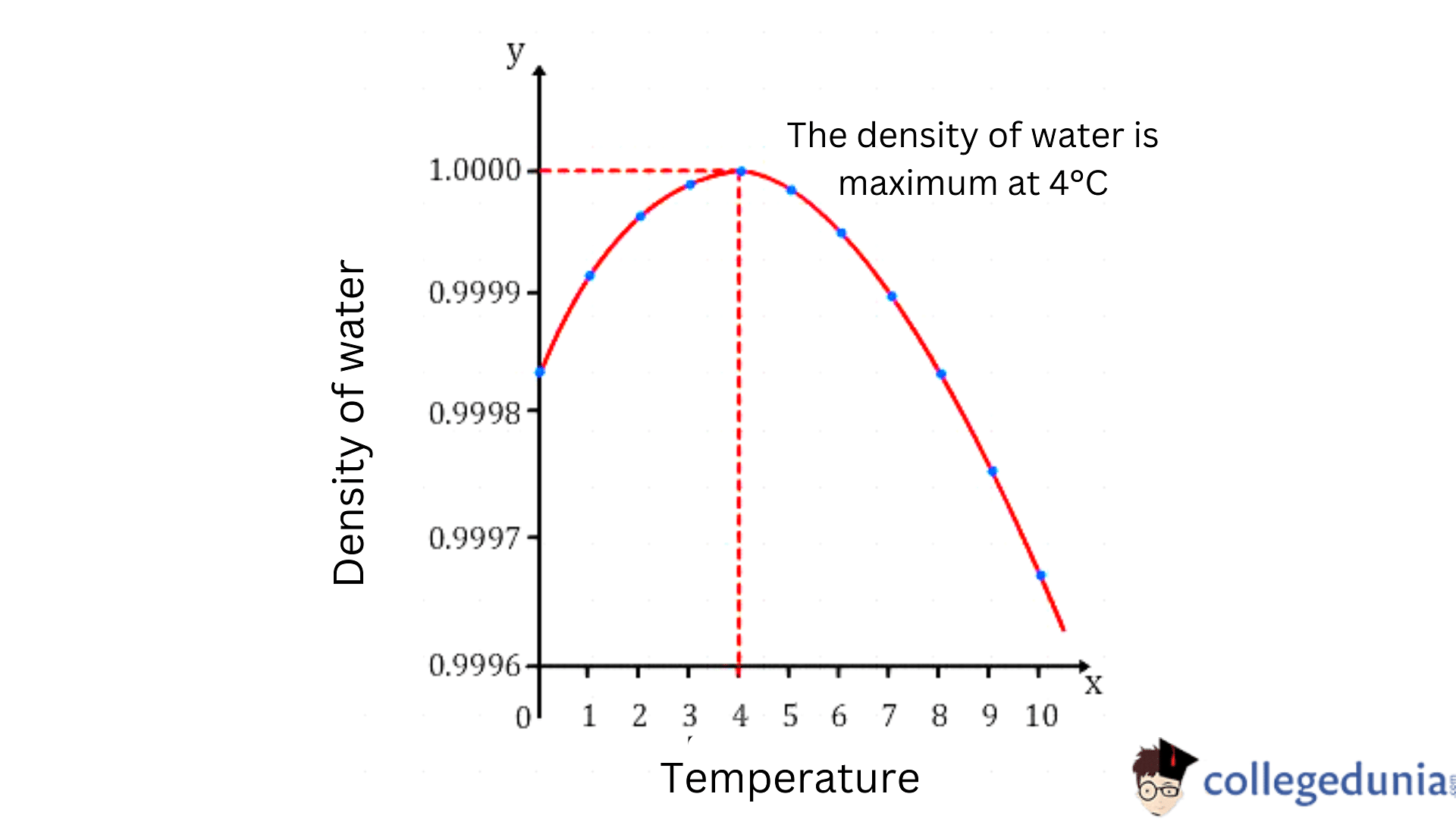
Get the free 'Water Density Calculator' widget for your website, blog, Wordpress, Blogger, or iGoogle. Heat required to convert 1 g of ice at 00C into steam at 1000C is ( LSteam 536cal/g ) : View Solution. Reason: The maximum density of water occurs at a temperature of 40C and after this temperature water expands on cooling. The Density of Water by Temperature calculator computes the density of water as a function of temperature, using the standard density of water (ρ) at standard temperature and pressure, and the unique temperature expansion coefficient of water. This widget calculates the density of water at various temperatures and pressures. Assertion :Water is denser than ice at same temperature.
